 
If mankind had been present at the beginning of the Earth, they would not have needed to enrich uranium, because the content of fissile 235U was significantly higher. The 0.72% observed today are only a residue caused by the difference in the half-lifes of 235U and 238U. At the time of the formation of the Earth, 235U was 85 times more abundant. In fact, 235U is the only existing fissile nucleus from naturally-occurring isotopes and therefore is a highly strategic material. 235U was the first isotope that was found to be fissile. 235U occasionally decays by spontaneous fission with very low probability of 0.0000000072%.Ģ35U is a fissile isotope, which means 235U is capable of undergoing fission reaction after absorbing thermal neutron. Moreover, 235U meets also alternative requirement that the amount ( ~2.43 per one fission by thermal neutron) of neutrons produced by fission of 235U is sufficient to sustain a nuclear fission chain reaction. 235U decays via alpha decay (by way of thorium-231) into 231Pa. For its very long half-life, it is still present in the Earth’s crust. 235U belongs to primordial nuclides, because its half-life is comparable to the age of the Earth (~4.5×10 9 years). This isotope has half-life of 7.04×10 8 years ( 6.5 times shorter than the isotope 238) and therefore its abundance is lower than 238U (99.28%). Uranium-235 has a half-life of 700 million years.Uranium 235, which alone constitutes 0.72% of natural uranium is the second common isotope of uranium in the nature. Most modern nuclear arsenals use plutonium as the fissile component, however U-235 devices remain a nuclear proliferation concern due to the simplicity of the design. The Little Boy atomic bomb was fueled by enriched uranium. However, judicious use of implosion and neutron reflectors can enable construction of a weapon from a quantity of uranium below the usual critical mass for its level of enrichment, though this would likely only be possible in a country which already had extensive experience in developing nuclear weapons. The fissile uranium in nuclear weapons usually contains 85% or more of 235U known as weapon(s)-grade, though for a crude, inefficient weapon 20% is sufficient (called weapon(s)-usable) even less is sufficient, but then the critical mass required rapidly increases. Uranium which has been processed to boost its uranium-235 proportion is known as enriched uranium, different applications require unique levels of enrichment. Pressurised Heavy Water Reactors, other heavy water reactors, and some graphite moderated reactors are known for using unenriched uranium. This concentration is insufficient for a self sustaining reaction in a light water reactor enrichment, which just means separating out the uranium-238, must take place to get a usable concentration of uranium-235. Only around 0.72% of all natural uranium is uranium-235, the rest being mostly uranium-238. For fast neutrons it is in the order of 1 barn. The nuclear cross section for slow thermal neutrons is about 1000 barns. However, approximately 5% of this energy is carried away by virtually undetectable neutrinos. The fission of one atom of U-235 generates 200 MeV = 3.2 × 10 -11 J, i.e. 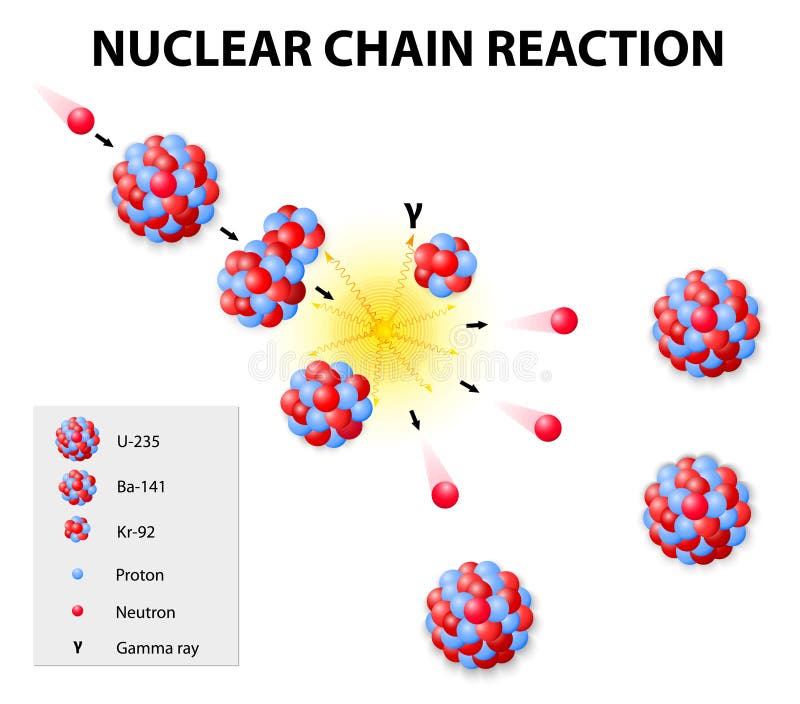
In nuclear bombs, the reaction is uncontrolled and the large amount of energy released creates a nuclear explosion. In nuclear reactors, the reaction is slowed down by the addition of control rods which are made of elements such as boron, cadmium, and hafnium which can absorb a large number of neutrons. Some of them produce neutrons, called delayed neutrons, which contribute to the fission chain reaction. A fission chain reaction produces intermediate mass fragments which are highly radioactive and produce further energy by their radioactive decay. A critical chain reaction can be achieved at low concentrations of U-235 if the neutrons from fission are moderated to lower their speed, since the probability for fission with slow neutrons is greater. If the reaction will sustain itself, it is said to be critical, and the mass of U-235 required to produce the critical condition is said to be a critical mass. If at least one neutron from U-235 fission strikes another nucleus and causes it to fission, then the chain reaction will continue. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
